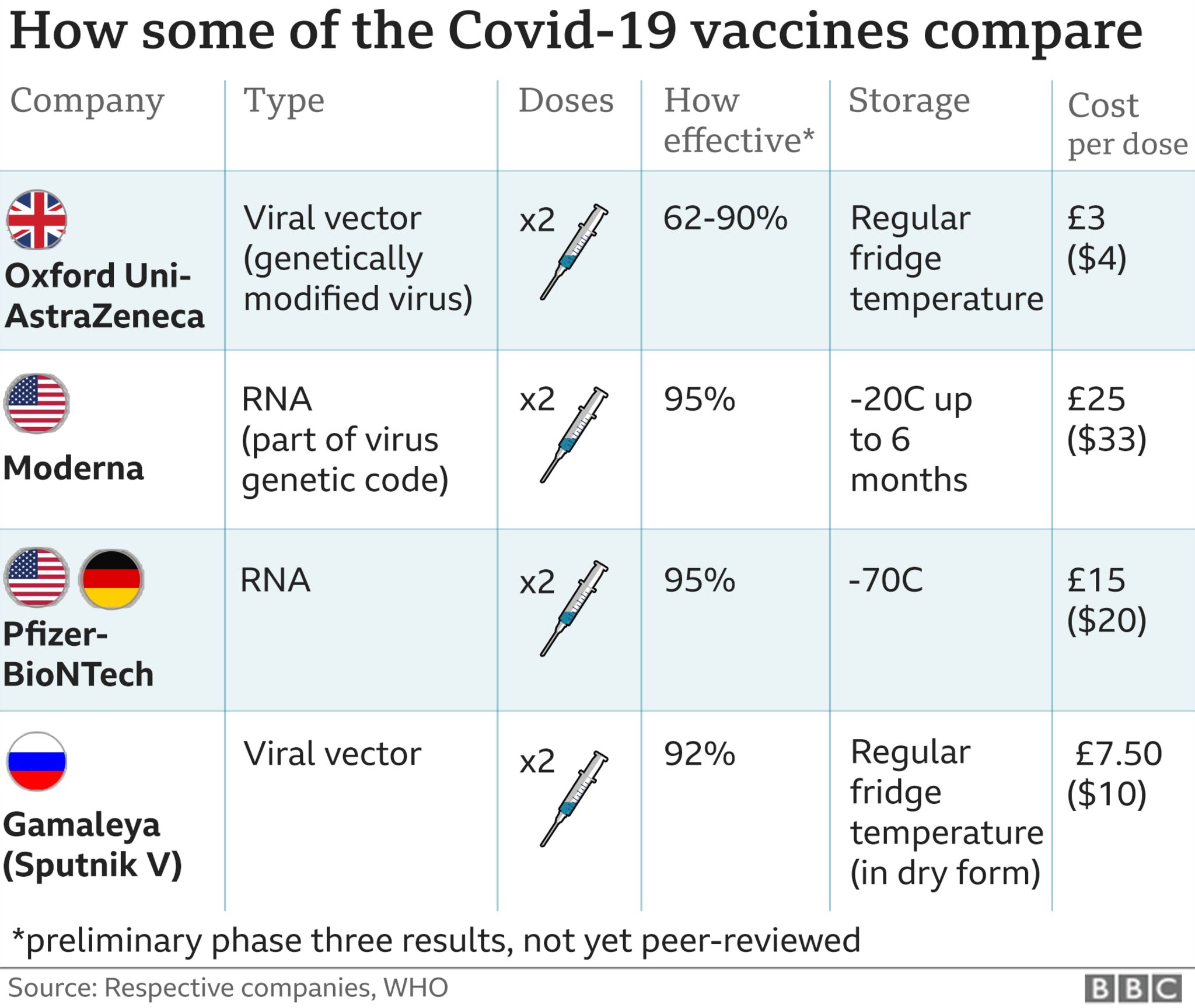
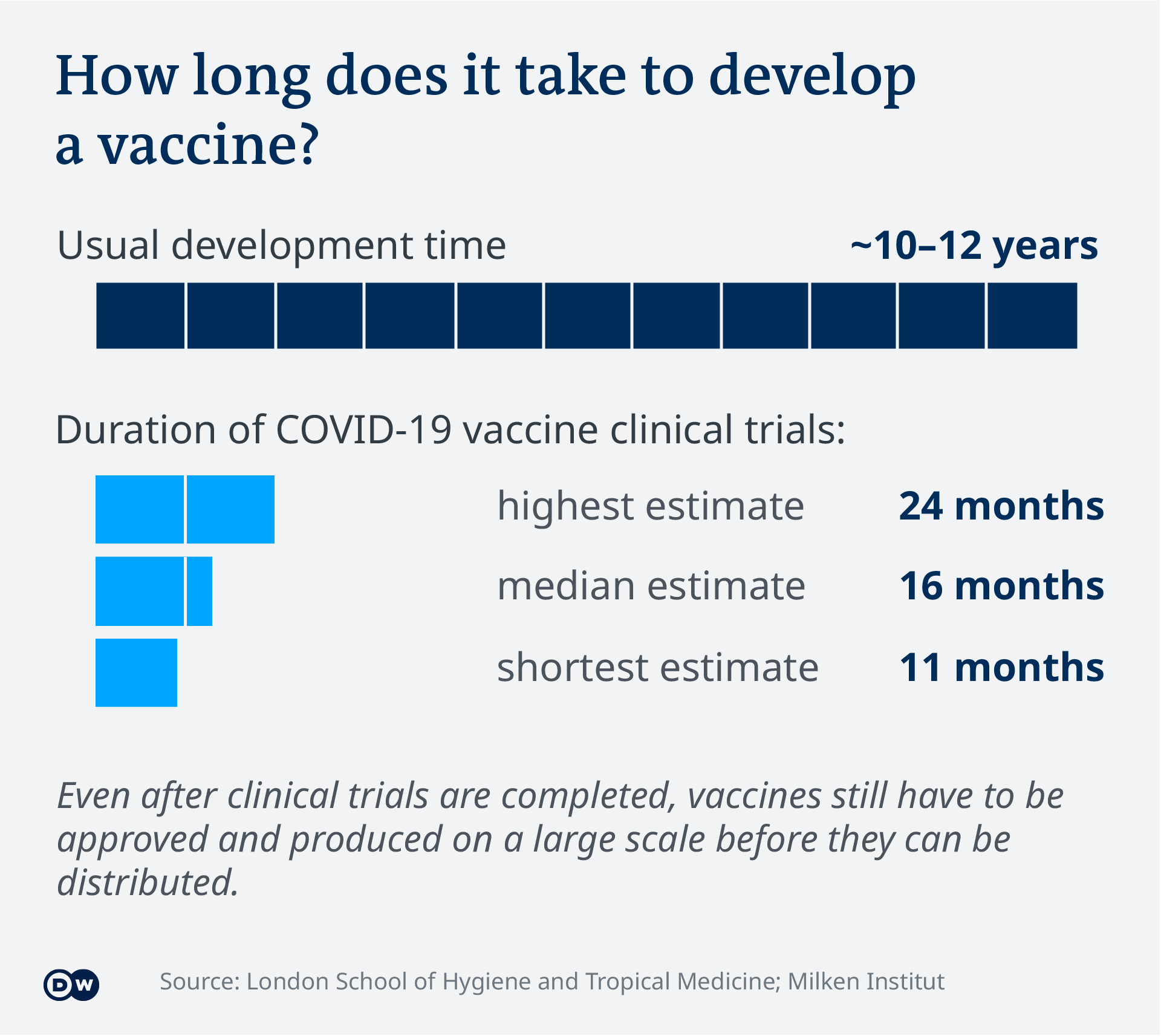
COVID-19 vaccines could become mandatory. Vaccine mandates are legal within specific parameters as affirmed by the US.
 Why The Covid 19 Vaccine Should Be Mandatory Opinion The Jakarta Post
Why The Covid 19 Vaccine Should Be Mandatory Opinion The Jakarta Post
It is not about the government overruling the individual preference for the risk of isolated impacts.

Is the vaccine mandatory. But as the US. The short answer is yes though different kinds of requirements apply in different situations. The legal precedent for mandatory vaccinations dates back to a 1905 Supreme Court case after a smallpox outbreak in Massachusetts.
In August Australias prime minister suggested the vaccine would be mandatory in his country but later backtracked on those comments. No vaccines are mandatory. That same month in Virginia the health commissioner said he.
Rutgers University is the first American college to make student vaccinations mandatory for on-campus courses. Heres how it might work. COVID-19 vaccination should be mandatory at least for certain groups.
States have the legal and constitutional authority to require that the people who live in that state be vaccinated or to introduce a vaccine mandate. It is unlikely the Covid vaccine could become mandatory for everyone in the UK as no other vaccine currently holds that legal status. As vaccine-preventable diseases surge governments around the world are putting down the carrots and bringing out bigger sticks in the form of mandatory vaccinations.
Reaches important clinical milestones in its vaccine development efforts questions arise relating to. But there are going to be individual institutions that Im. 5 Contagiousness and method of transmission.
At Baylor College of Medicine in Houston some 14000 employees and students are required to get the annual flu vaccine says Dr. Compulsory vaccination of college students may. There is no place for selfish.
The Public Health Control of Disease Act 1984 which applies. An employer can establish a mandatory vaccination policy if the need for it is job-related or if remaining unvaccinated would pose a direct threat. Since the 2009 swine flu pandemic the Equal Employment Opportunity Commission has held that employers are allowed to.
Hajdu has been explicit in saying when a COVID-19 vaccine is made available it will not be mandatory. When the Supreme Court Ruled a Vaccine Could Be Mandatory A 1905 decision provided a powerful and controversial precedent for the flexing of government authority. Private vaccine mandates have broadly been considered legal.
The programs seem to be working but a recent report points out it isnt without cost. Federally no according to Health Minister Patty Hajdu. The short answer is yes.
Vaccines against diseases that are easy to catch for instance by walking into a room with another infected person are good candidates for being mandatory. The health secretary tells Sky News the issue is being looked into by. This means there would be penalties for failure to vaccinate such as fines or limitations on freedom of movement.
Mandatory vaccination deals with social welfare maximization. The average person is never likely to encounter either virus so vaccination makes no sense let alone mandatory vaccination. Vaccines could be made mandatory for care workers says Matt Hancock.
After a COVID-19 vaccine is available you may need to get inoculated to go to. Im not sure its the COVID-19 vaccine going to be mandatory from a central government standpoint like federal government mandates.